Turning the years from 2020 to 2021 herald in a time of hope with many health authorities worldwide, including Singapore, announcing vaccination plans for COVID-19. In his televised address on 14 December 2020, Singapore Prime Minister Lee Hsien Loong said, “Now that vaccines are becoming available, we can see light at the end of the tunnel.” [1] Singapore will offer vaccinations to the entire adult population at no cost, voluntarily. Progressive vaccination is currently underway, and the hope is to cover everyone who wants a vaccination by the end of 2021. In countries with severe COVID-19 outbreak in the community such as Britain and America, vaccination has already started in early December 2020.

Given the voluntary nature of the vaccination, many people are pondering the same question, “Should I?” To make an informed decision, one must understand what the vaccines are, what the world has learned about the effectiveness and risk of these vaccines so far, and the unknowns.
Pfizer-BioNTech and Moderna – What are they?
In Singapore, the Pfizer-BioNTech (BNT162b2) vaccine is the first to roll out, and the Moderna (mRNA-1273) vaccine will also be arriving soon. Both vaccines are based on the nucleoside-modified messenger Ribonucleic acid (mRNA) technology. They are synthetic mRNA encoded with part of the spike protein found on the surface of the SARS-CoV-2 coronavirus. Once injected into the body, these mRNA will trigger the cells to make spike protein fragments similar to the COVID-19 virus. Since these protein fragments are not the real virus, they pose no harm to the body. However, the immune cells will detect these protein fragments as foreign “invaders” and produce antibodies to target and destroy them. In this way, the body learned to defend against COVID-19 virus without acquiring the actual disease.

How effective are these vaccines?
Pfizer-BioNTech is said to be 95% effective. How does this number come about, and what does it mean? The 95% effective rate was reported by the scientists who conducted the vaccine’s clinical trial in The New England Journal of Medicine (NEJM), one of the world’s most prestigious medical journals on 10 December 2020 [2]. Forty-three thousand four hundred forty-eight (43,448) participants age 16 and above were randomly assigned to receive injections of either the vaccine or a saline placebo. Each participant received two 0.3 ml doses of injections on the arm, 21 days apart. Participants were followed up to detect for any symptoms of COVID-19 after seven days from the second injection. By 9 October 2020, 8 Covid-19 cases out of 18,198 who received the vaccine and 162 cases in the placebo group of 18,325 were reported [2]. Thus, the number of cases in the vaccine group is 95% less than the placebo group, which is how the 95% effective rate is derived.
Similarly, the Moderna trial results were reported in the NEJM on 30 December 2020 [3]. A total of 185 cases of COVID-19 were found in the placebo group (Total: 14,073), whereas only 11 were reported in the vaccine group (Total: 14,134) [3]. Hence, in comparison, those who received the vaccine reported 94.1% fewer cases of COVID-19.
| Vaccine type | Vaccine group | Placebo group | Ratio | Effectiveness |
|---|---|---|---|---|
| Pfizer-BioNTech | 8 / 18,198 (4.4 per 10,000) | 162 / 18,325 (88.4 per 10,000) | 1 : 20.1 (0.5%) | 95% |
| Moderna | 11 / 14,134 (7.8 per 10,000) | 185 / 14,073 (131.5 per 10,000) | 1 : 16.9 (0.6%) | 94% |
The results reported from both trials are considered interim only since both are still on-going.
Are there any side-effects from the vaccines?
The safety data from both trials showed that the participants who received the vaccines reported more side-effects than those who received placebo. About 66% of Pfizer-BioNTech vaccine recipients had mostly mild pain at the injection sites with very few developed redness and swelling [2]. For the Moderna vaccine, 86% of recipients had pain at the injection sites [3]. Nevertheless, the painful sensations are mostly mild. Other uncomfortable effects include fever, chills, headache, fatigue, muscle pain, joint pain, nausea or vomiting, and diarrhea [2,3]. Although most of these events were mild, some participants did report severe reactions. Nonetheless, both trials did not have participants that die due directly to the side-effects [2,3].

One adverse reaction that was not reported in these studies but was subsequently documented in during the roll-out of the vaccines was severe allergic reactions or anaphylaxis. The Centers of Disease Control and Prevention in the United States, or more commonly known as CDC, issued a preliminary finding that between 14 to 23 December 2020, there were 21 cases of anaphylaxis after administration of the first 1,893,360 doses of the Pfizer-BioNTech COVID-19 vaccine (11.1 cases per million doses). About 71% of these events occurred within 15 minutes of vaccination [4]. Cases of anaphylaxis due to Pfizer-BioNTech vaccine were also reported in the United Kingdom. With this, the British health authority had issue advice that anyone with a history of anaphylaxis to a vaccine, medicine or food should not receive the Pfizer-BioNTech vaccine [5]. As for the Moderna vaccine, there is no report of any severe allergic reaction to date.
What are the unknowns?
Although both vaccines’ effective rates are impressive based on the initial data reported, there are still many unknowns. Firstly, the observation periods of the preliminary reports from both trials are short, only about two to three months. We still do not know how long the vaccines’ effect will sustain and whether there is a need to re-vaccinate periodically. Secondly, there is no data on any potential long-term side-effects of the vaccines; only time can tell. Thirdly, coronavirus is known to mutate quickly. Hence, the spike protein of COVID-19 coronavirus could potentially mutate out recognition. How fast the COVID-19 coronavirus can mutate is still anyone’s guess [6]. Lastly, even if the vaccines can stay effective for a more extended period, a substantial proportion of the population will need to be inoculated to achieve herd immunity against COVID-19 safely. Experience from other infectious diseases shown that it can be anything from 80% to 95%. Hence, the percentage of the population that needs to be vaccinated to curb COVID-19 successfully is unknown.

To be or not to be?
Around the world, with the COVID-19 pandemic is still raging. Thus, a call must be made on when to roll out the vaccines to contain the spread. Most experts and health authorities agree that the vaccines’ benefits outweigh their potential risks after considering the available data. Thus, the vaccines are approved for use in the larger population, even though many unknowns still exist.
I have already heard about people discussing the story of death due to anaphylaxis even in the coffee shop, not to mention over social media. It is known that false or negative news spread much faster over social media than facts [7]. The public should not be overly concerned with the possibility of vaccine anaphylaxis. Severe allergic reaction to COVID-19 vaccines is not unique. The risk of anaphylaxis across all different types of vaccines is estimated to be around 1.31 per million vaccine doses. Some vaccines have a higher rate of hypersensitivity than others. For example, the Measles vaccine had recorded as high as 120 cases per million vaccine doses.
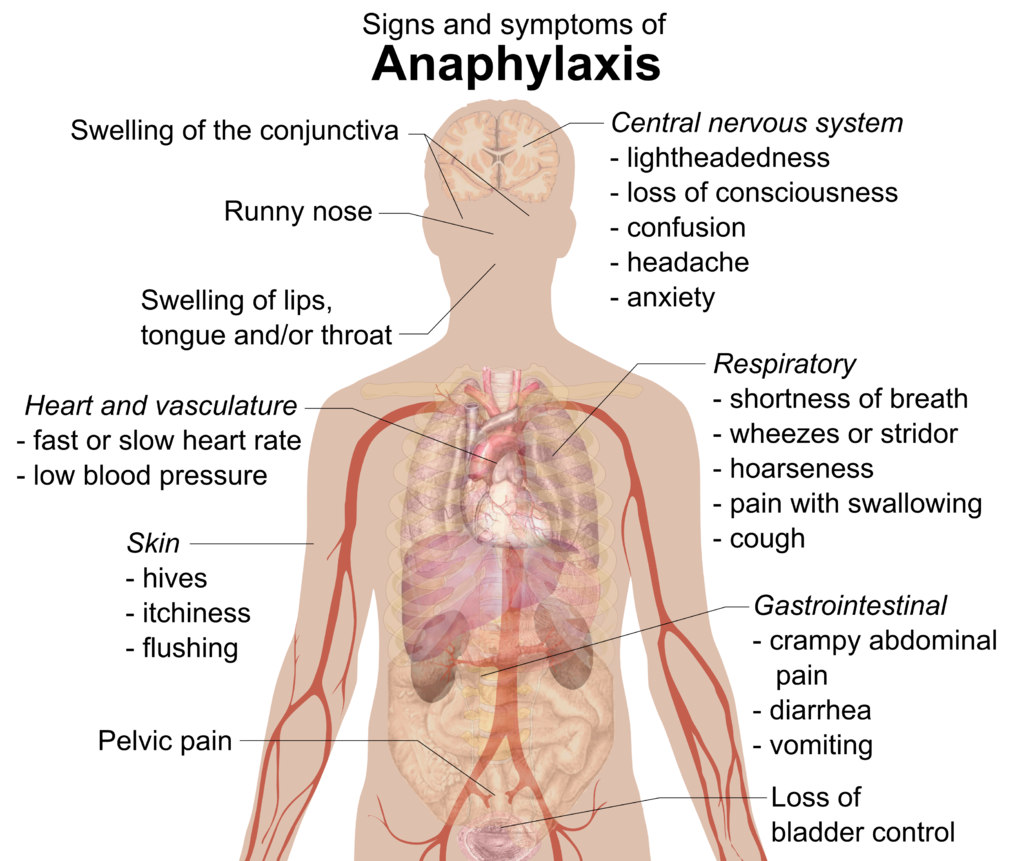
In contrast, the combination MMR (measles, mumps, and rubella) vaccine accounted for 12.5 cases per million vaccine doses. In comparison, the 11.1 cases per million doses observed with Pfizer-BioNTech vaccine should not be a significant concern [8]. With proper monitoring after vaccination, any hypersensitivity reaction can be quickly addressed by trained health care personnel immediately.
Given that the percentage population that needs to be vaccinated is unknown, for Singapore, the strategy is to quickly vaccinate as many people as possible to achieve the community’s necessary immunity. The shots will be given free, but people are given a choice. Hence, taking the vaccine shots should weigh between individual fear/belief and the public good. Give the evidence, the associated risk, and the unknowns, what is your decision?
Conclusion
Progressive vaccination is currently underway in Singapore and many countries. To decide whether to take up the vaccine shots, one should objectively evaluate the information from trusted sources. One should understand the evidence, the risk, and the unknowns before making an informed decision.
References
[1] H.L. Lee, We are seeing light at the end of the tunnel: PM Lee, Opinion News & Top Stories – The Straits Times, The Straits Times. (2020). https://www.straitstimes.com/opinion/we-are-seeing-light-at-the-end-of-the-tunnel-pm-lee (accessed January 14, 2021).
[2] F.P. Polack, S.J. Thomas, N. Kitchin, J. Absalon, A. Gurtman, S. Lockhart, J.L. Perez, G. Pérez Marc, E.D. Moreira, C. Zerbini, R. Bailey, K.A. Swanson, S. Roychoudhury, K. Koury, P. Li, W. V. Kalina, D. Cooper, R.W. Frenck, L.L. Hammitt, Ö. Türeci, H. Nell, A. Schaefer, S. Ünal, D.B. Tresnan, S. Mather, P.R. Dormitzer, U. Şahin, K.U. Jansen, W.C. Gruber, Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine, N. Engl. J. Med. 383 (2020) 2603–2615. doi:10.1056/nejmoa2034577.
[3] L.R. Baden, H.M. El Sahly, B. Essink, K. Kotloff, S. Frey, R. Novak, D. Diemert, S.A. Spector, N. Rouphael, C.B. Creech, J. McGettigan, S. Khetan, N. Segall, J. Solis, A. Brosz, C. Fierro, H. Schwartz, K. Neuzil, L. Corey, P. Gilbert, H. Janes, D. Follmann, M. Marovich, J. Mascola, L. Polakowski, J. Ledgerwood, B.S. Graham, H. Bennett, R. Pajon, C. Knightly, B. Leav, W. Deng, H. Zhou, S. Han, M. Ivarsson, J. Miller, T. Zaks, Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine, N. Engl. J. Med. (2020) 1–14. doi:10.1056/nejmoa2035389.
[4] CDC COVID-19 Response Team; Food and Drug Administration, Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine — United States, December 14–23, 2020, MMWR Morb Mortal Wkly Rep. (2021). doi:10.15585/mmwr.mm7002e1.
[5] Managing allergic reactions to Pfizer-BioNTech COVID-19 vaccine, React. Wkly. 1836 (2021) 5. doi:10.1007/s40278-021-87824-3.
[6] M.F. Haidere, Z.A. Ratan, S. Nowroz, S. Bin Zaman, Y.-J. Jung, H. Hosseinzadeh, J.Y. Cho, COVID-19 Vaccine: Critical Questions with Complicated Answers, Biomol. Ther. (Seoul). 29 (2021) 1–10. doi:10.4062/biomolther.2020.178.
[7] S. Vosoughi, D. Roy, S. Aral, The spread of true and false news online, Science (80-. ). 359 (2018) 1146 LP – 1151. doi:10.1126/science.aap9559.
[8] M.M. McNeil, F. DeStefano, Vaccine-associated hypersensitivity, J. Allergy Clin. Immunol. 141 (2018) 463–472. doi:10.1016/j.jaci.2017.12.971.